Jika Ksp Ag2CrO4 = 4 x10^12, kelarutan Ag2CrO4 da...
Calculate the solubility of Ag2CrO4 (Ksp = 1.31 x 10-12) a) in pure H2O b) in 0.0100 M AgNO3 c) in 0.0100 MNa2CrO4. 6 comments. Upgrade to add a comment. BP Brendan P. March 12, 2023. the answer provided seems to be option b which is 1.4410-8! but the explanation is, like kinda hard to follow could be clearer! RB.

The Ksp of Ag2CrO4 is 1.1 × 10^12 at 298 K . The solubility (in mol L^1 ) of Ag2CrO4 in a 0.1
Ksp = 1.12x10-12. Ksp = [Ag +] 2 [CrO 4 2-] The common ion would be CrO 4 2-since you have 1.20 M potassium chromate (K 2 CrO 4). Because the [CrO 4 2-] is so large from the potassium chromate (1.2 M) we can ignore the [CrO 4 2-] contributed by the Ag 2 CrO 4 as it will be very small. Now we can write the Ksp expression as follows: Ksp = [Ag.
Ag2CrO4 and Ag2C2O4 both are present in a saturated solution in water .Ksp of Ag2CrO4 and
Pembahasan. Hasil Kali Kelarutan (Ksp) adalah hasil kali konsentrasi ion-ion dari larutan jenuh garam yang sukar larut dalam air. Setelah itu, masing-masing konsentrasi dipangkatkan dengan koefisien menurut persamaan ionisasinya. Persamaan reaksi ionisasi garam adalah sebagai berikut. Rumus Ksp untuk persamaan reaksi di atas adalah .

The Ksp of Ag2CrO4, AgCl, AgBr and Agl are respectively, 1.1 × 10^12, 1.8 × 10^10 , 5.0 × 10
Contoh Soal Kelarutan dan Hasil Kali Kelarutan (KSP) dan Pembahasan Contoh Soal 1: Hitunglah kelarutan Cu(OH) 2 dalam satuan g/L, jika diketahui K sp Cu(OH) 2 = 2,2 × 10 −20. Pembahasan: Contoh Soal 2: Hitunglah kelarutan molar PbI 2 dalam larutan KI 0,1 M. (K sp PbI 2 = 7,1 × 10 −9) Pembahasan: Dalam larutan, KI akan terdisosiasi menjadi.

Rumus hasil kali kelarutan (KSP) Ag2CrO4 dinyatakan sebag...
The [Ag+] in a saturated solution of Ag2CrO4 is 4 x 10−4 mol/L. Find Ksp of Ag2CrO4. The K s p of A g 2 C r O 4 is 1.1 × 10 − 12 at 298 K. The solubility (in mol L − 1) of A g 2 C r O 4 in a 0.1 M A g N O 3 solubility is: The ksp of Ag2CrO4 is 1.1 ×10−12 at 298k.

The Ksp of Ag2CrO4 = 1.2 × 10^11 . What concentration Ag^+ ion in aqueous solution will just
The equilibrium constant for a dissolution reaction, called the solubility product ( Ksp ), is a measure of the solubility of a compound. Whereas solubility is usually expressed in terms of mass of solute per 100 mL of solvent, Ksp is defined in terms of the molar concentrations of the component ions. In contrast, the ion product ( Q) describes.

SOLVED The Ksp of silver chromate (Ag2CrO4) is 1.12 X1012 (a) What is the solubility of the
Silver chromate is a chemical compound of silver and hexavalent chromium. It is used in photography. Hexavalent chromium refers to chemical compounds that contain the element chromium in the +6 oxidation state. Chromium (VI) is more toxic than other oxidation states of the chromium atom because of its greater ability to enter cells and higher.

The Ksp of Ag2CrO4 , AgCl, AgBr and AgI are respectively, 1.1 × 10^12, 1.8 × 10^10, 5.0 × 10
Jika kelarutan Ag2CrO4 dalam air adalah 5mol/Liter , tentukan hasil kali kelarutan Ag2CrO4 ! Jawab : Ag2CrO4 ⇔ 2Ag+(aq) + CrO4-2(aq) s 2s s Ksp Ag2CrO4 = […

Given the two standard reduction potentials below what is the ksp of ag2cro4 at 25 °c
Question: Which is the correct Ksp expression for Ag2CrO4 (s) dissolving in water? Ksp = [Ag*12(CrO42-1 Ksp = (Ag*12[Cr3+][02-14 Ksp = [A8+][Cro_2-12 Ksp - [Ag*][Cro42-1 . Show transcribed image text. There are 2 steps to solve this one. Who are the experts?

The Ksp of Ag2CrO4 is 1.1×10−12 at 298 K. The solubility (mol/L) of Ag2CrO4 in 0.1MAgNO3
M x A y (s) → x M y+ + y A x-. Karena zat pada tidak mempunyai molaritas, maka tetapan kesetimbangan reaksi diatas hanya melibatkan ion-ionnya saja, dan tetapan kesetimbangannya disebut tetapan hasil kali kelarutan (Ksp).. Ksp = [M y+] x [A x-] y. Hubungan Kelarutan dengan Ksp. Secara umum, hubungan antara kelarutan (s) dengan tetapan hasil kali kelarutan (Ksp) untuk larutan elektrolit A x B.
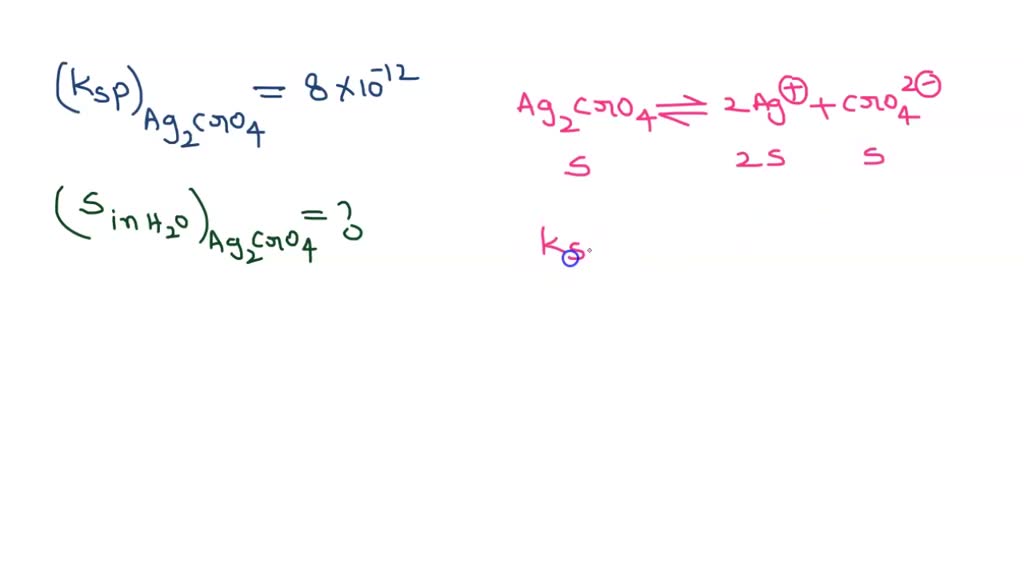
SOLVED Jika ksp Ag2CrO4 adalah 4x 10pangkat12 , kelarutan Ag2CrO4 (Mr= 332) dalam air adalah
Homework Statement I have to calculate the KSP of Ag2CRo4. I did this lab several days ago and I'm completely stumped. If anyone could help me out here, at the least guide me in the right direction so I can get this done as I am getting very frustrated :P Here is the data I have derived thus far. 2.5x10-5 = concentration of AgNO 3 1.5x10-5 = concentration of K 2 CrO 4 Using a concentration.
the ksp of AgCrO4 is 1.1 ×10^ 12 at 298K. the solubility of Ag2CrO4 in a 0.1 M AgNO3 solution is?
Rumus hasil kali kelarutan (KSP) Ag2CrO4 dinyatakan sebag… from colearn.id Pendahuluan. Kimia adalah ilmu yang mempelajari tentang struktur, sifat, dan reaksi dari materi. Salah satu konsep penting dalam kimia adalah kesetimbangan kimia. Kesetimbangan kimia terjadi ketika reaksi berbalik dan maju dengan kecepatan yang sama.

Rumus hasil kali kelarutan (KSP) Ag2CrO4 dinyatakan sebag...
Halo friend ada pertanyaan mengenai kelarutan dan hasil kali kelarutan Nah kita disuruh untuk mencari rumus hasil kali kelarutan atau KSP ag2 cro4 AG 2 cro4 ini akan terurai menjadi 2 AG plus ditambah cro4 2min untuk kelarutan ag2 cro4 yaitu untuk AG + yaitu 2s dan untuk cro4 2min adalah es untuk menentukan KSP nya yaitu larutan dari AG + berpangkat 2 sesuai dengan koefisien kali kelarutan.

The precipitate of Ag2Cro4 (ksp=1 = 1x10... Physical Chemistry
Silver chromate is an inorganic compound with formula Ag 2 CrO 4 which appears as distinctively coloured brown-red crystals. The compound is insoluble and its precipitation is indicative of the reaction between soluble chromate and silver precursor salts (commonly potassium/sodium chromate with silver nitrate). This reaction is important for two uses in the laboratory: in analytical chemistry.

The Ksp for a sparingly soluble Ag2Cro4, is4 * 1012. The molar solubility of the salt is(1) 2.0
Ag2CrO4(s) ⇌ 2Ag+(aq) + CrO4^(2-) Rumus Ksp untuk Ag2Cro4 adalah: Ksp = [Ag+]^2[CrO4^2-] Dimana [Ag+] adalah konsentrasi ion perak (silver) dan [CrO4^2-] adalah konsentrasi ion kromat (chromate) dalam larutan jenuh. Cara Mengekspresikan Hasil Kali Kelarutan (Ksp) Ag2CrO4. Setelah kita memiliki nilai Ksp untuk Ag2CrO4, kita perlu.

Jika diketahui Ksp Ag2CrO4=4 x 10^12 , konsentrasi ion
The Ksp of Ag2CrO4 is 1.12× 10^-12. What is the solubility (in mol/L) of silver chromate in 1.50 M potassium chromate aq solution? In 1.50 M silver nitrate aq solution? In pure water? I'm assuming I need to write the solubility expression. If I have this right, Ksp=[2Ag]^2[CrO4] which becomes 1.12x10^-12=4s^3. But how to I then incorporate.