How Many Valence Electrons Are in the Neon Family
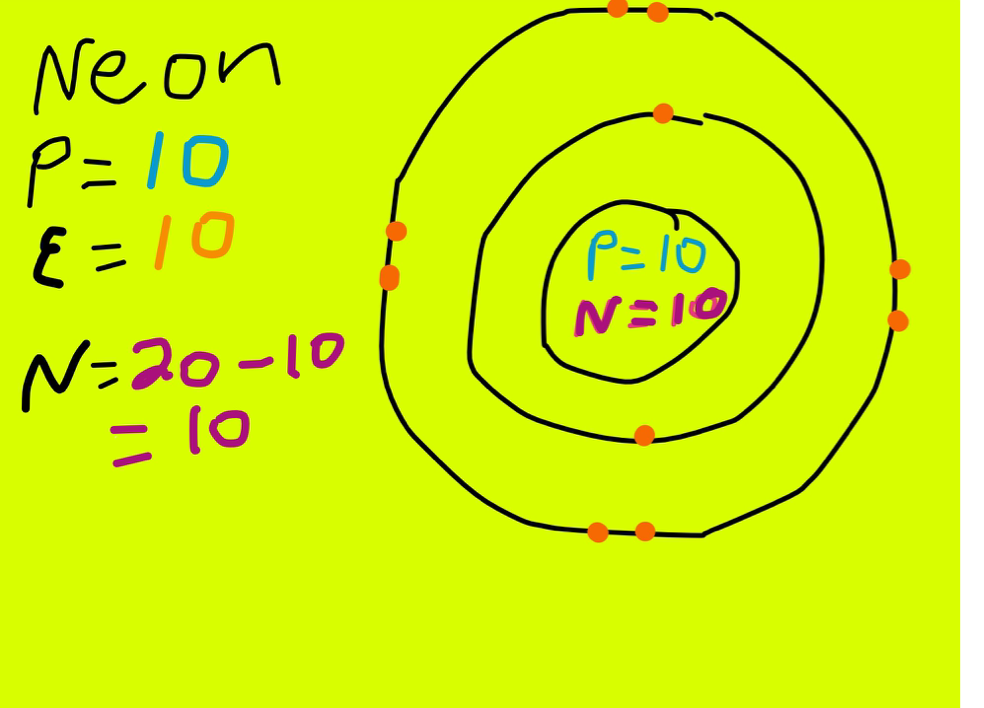
There are four simple steps to find out the valence electrons for neon atom which are: Step 1: Find the Atomic Number To find out the atomic number of neon, we can use the periodic table. With the help of the periodic table, we can easily see that the atomic number of neon is 10.
Neon Electron Configuration YouTube
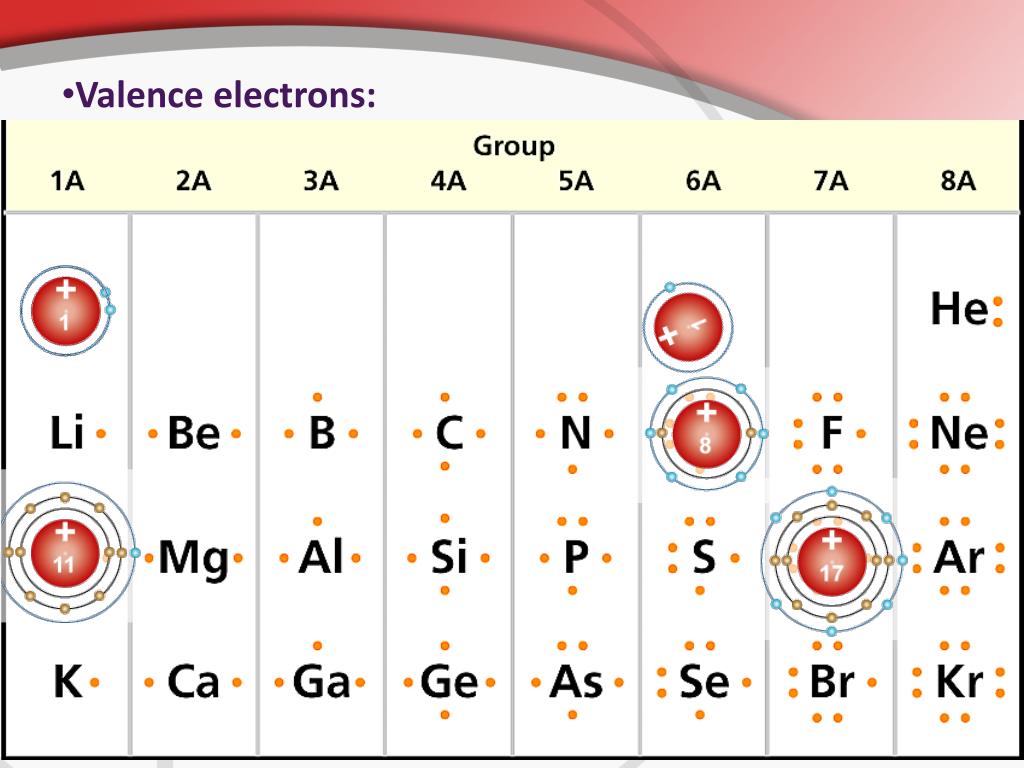
Each element has a number of valence electrons equal to its group number on the Periodic Table. Figure %: The periodicity of valence electrons This table. Helium (He) and Neon (Ne) have outer valence shells that are completely filled, so neither has a tendency to gain or lose electrons. Therefore, Helium and Neon, two of the so-called Noble.

How many valence electrons does neon(Ne) have?
How many valence electrons does neon have? Flexi Says: Valence electrons are the electrons in the outer energy level of an atom that can participate in interactions with other atoms. Each element has different number of valance electrons. The number of valence electrons in an atom is reflected by its position in the periodic table of the elements.
Electron Configuration Of Neon My XXX Hot Girl
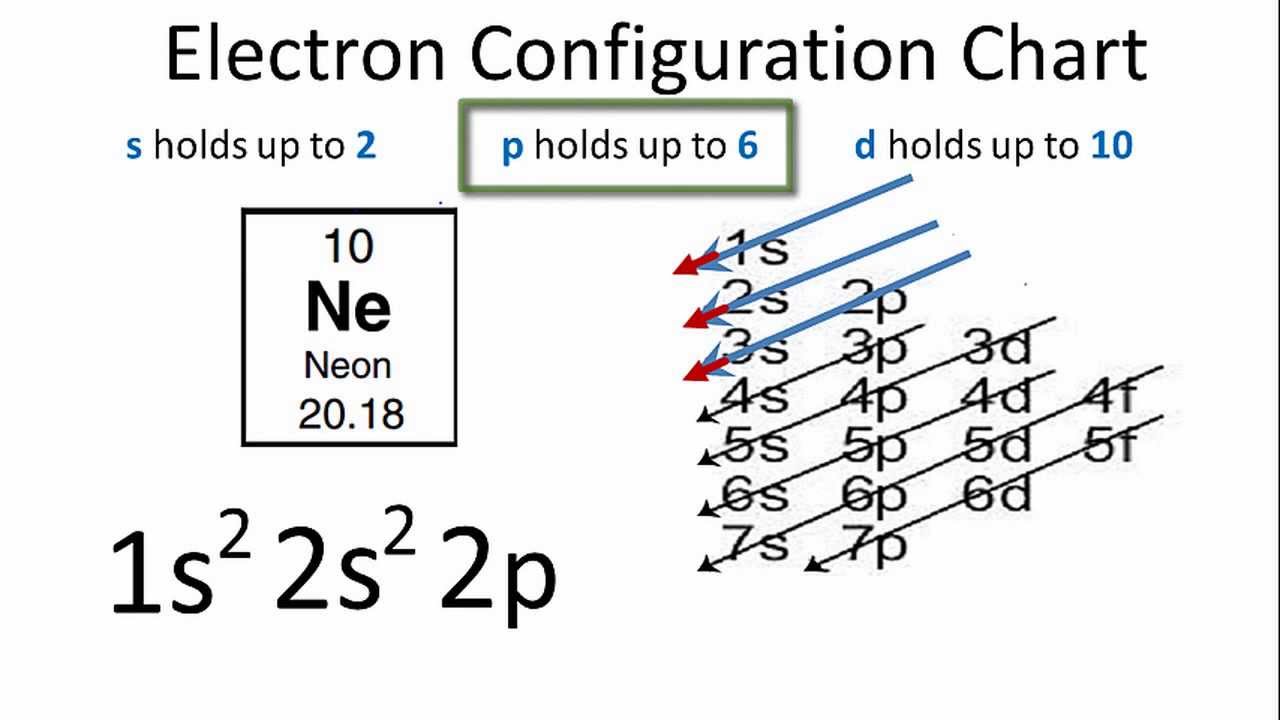
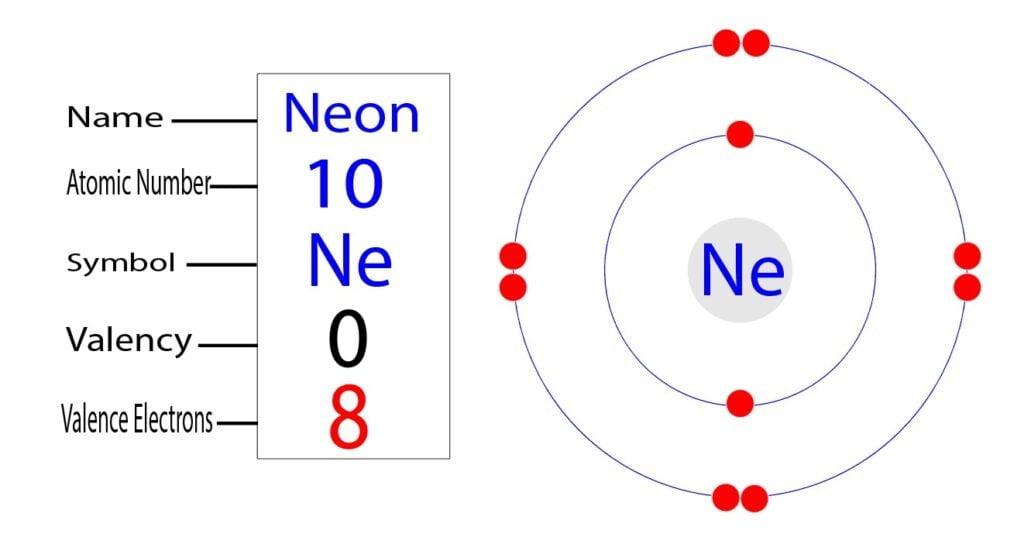
It simply depicts the theory that the first two orbital the 1s and the 2s are having 2 electrons respectively. The remaining 6 electrons have been held by the 2p orbital. How Many Valence Electrons Does Neon Have? Neon basically has the 8 valence electrons as its full octet and this is what makes neon stable in the terms of a noble gas.
How many valence electrons does sulfur(S) have?
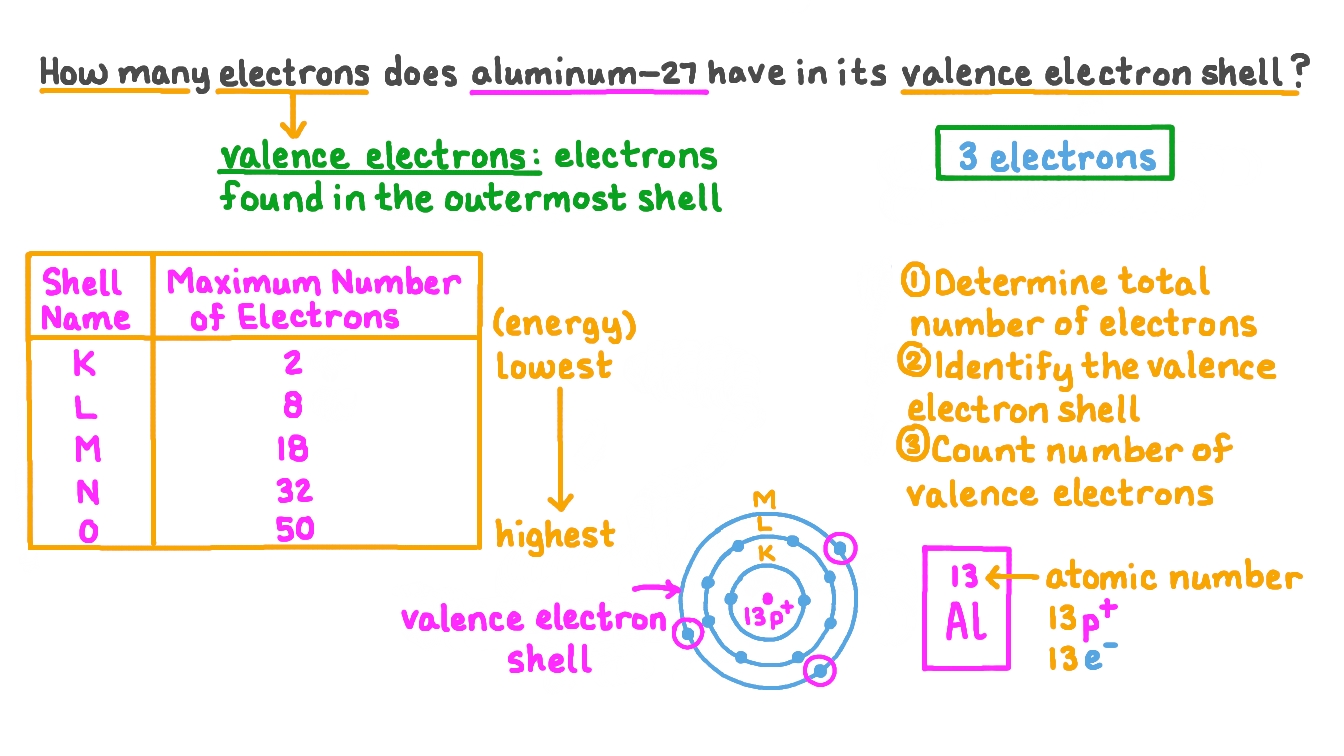
Valence electrons are the electrons present in the outermost shell of an atom. You can easily determine the number of valence electrons an atom can have by looking at its Group in the periodic table. For example, atoms in Groups 1 and 2 have 1 and 2 valence electrons, respectively. Atoms in Groups 13 and 18 have 3 and 8 valence electrons.
Question Video Determining the Number of Electrons in the Valence
How many valence electrons does boron have? Recognize that the second principal energy level consists of both the \(2s\) and the \(2p\) sublevels, and so the answer is three.. Neon, with its configuration ending in \(2s^2 2p^6\), has eight valence electrons. Valence electrons for transition elements. Transition elements are a bit trickier.
PPT Unit 3 PowerPoint Presentation, free download ID5685070
Please enable JavaScript to access the full features of the site. Glossary Allotropes Some elements exist in several different structural forms, called allotropes. Each allotrope has different physical properties. For more information on the Visual Elements image see the Uses and properties section below. Move to Fluorine Move to Sodium > Neon

What Are Valence Electrons? Definition and Periodic Table 8th Grade
Step-1: Determining the total number of electrons in neon First we need to know the total number of electrons in the neon atom. To know the number of electrons, you need to know the number of protons in neon. And to know the number of protons, you need to know the atomic number of the neon element. Position of neon (Ne) in the periodic table
5+ orbital diagram for neon RoomilaDeejay
Step 1) Figure out how many electrons the molecule must have. Carbon has 4 valence electrons. Each oxygen has 6 valence electrons. The -2 charge means that there are 2 extra electrons. Total: 4 + (3 × 6) + 2 = 24 electrons. The final answer MUST have this number of electrons‼!
Valency of Selenium Archives Dynamic Periodic Table of Elements and
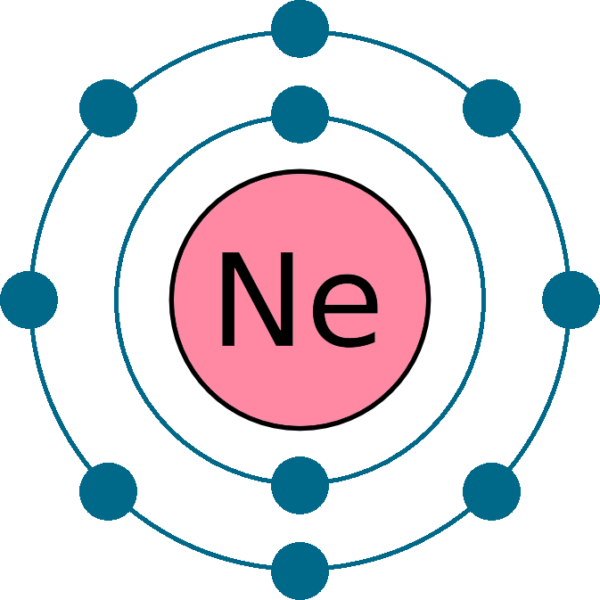
The Bohr model shows the atom as a central nucleus containing protons and neutrons, with the electrons in circular electron shells at specific distances from the nucleus, similar to planets orbiting around the sun.

How Many Valence Electrons Does Neon(Ne) Have?
There are two ways to find the number of valence electrons in Neon (Ne). The first is to use the Periodic Table to figure out how many electrons Neon has in its valence shell. To do.
Quia Electrons and the Periodic Table
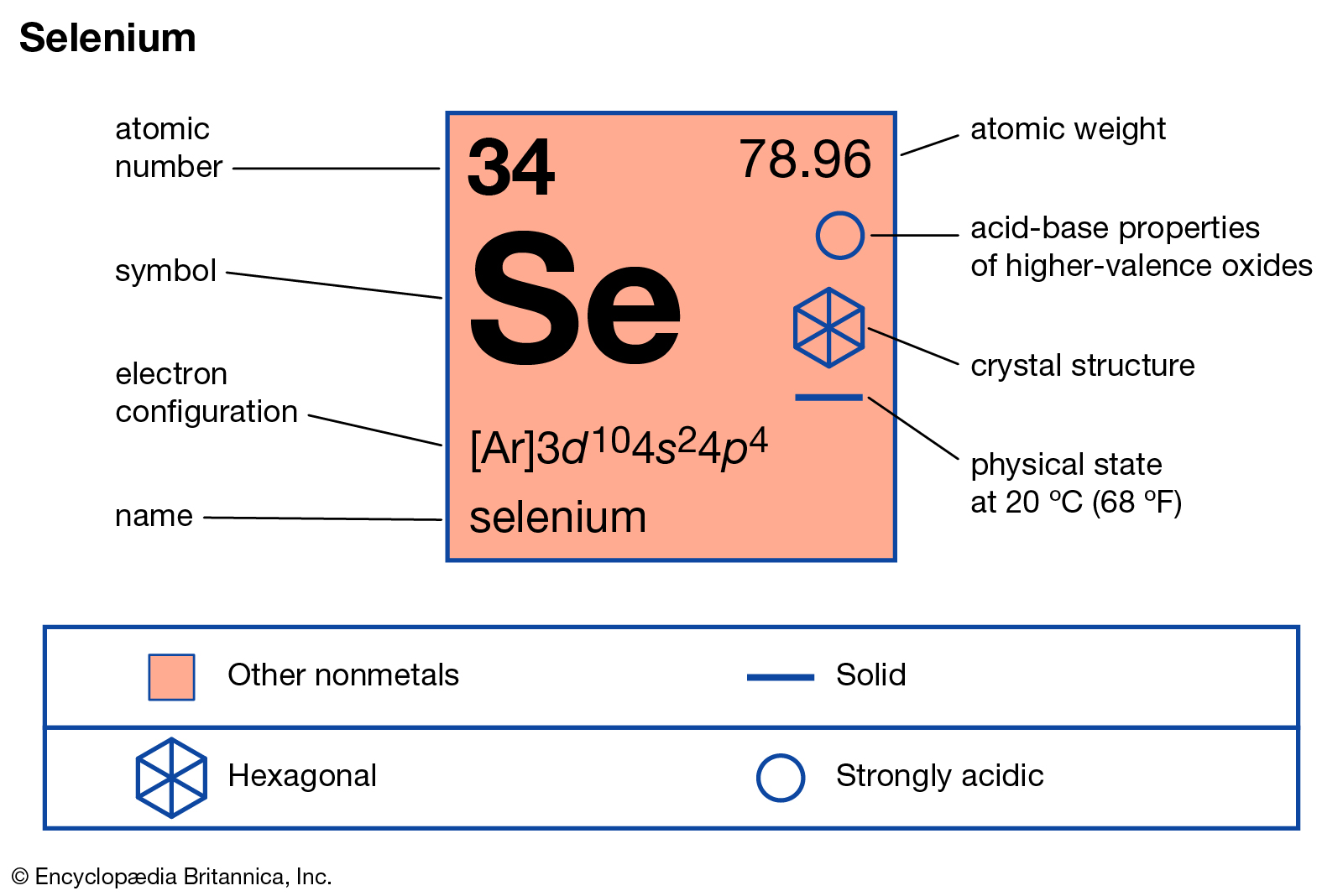
The arrangement of electrons in neon in specific rules in different orbits and orbitals is called the electron configuration of neon. The electron configuration of neon is [ He] 2s 2 2p 6, if the electron arrangement is through orbitals. Electron configuration can be done in two ways. Electron configuration through orbit (Bohr principle)
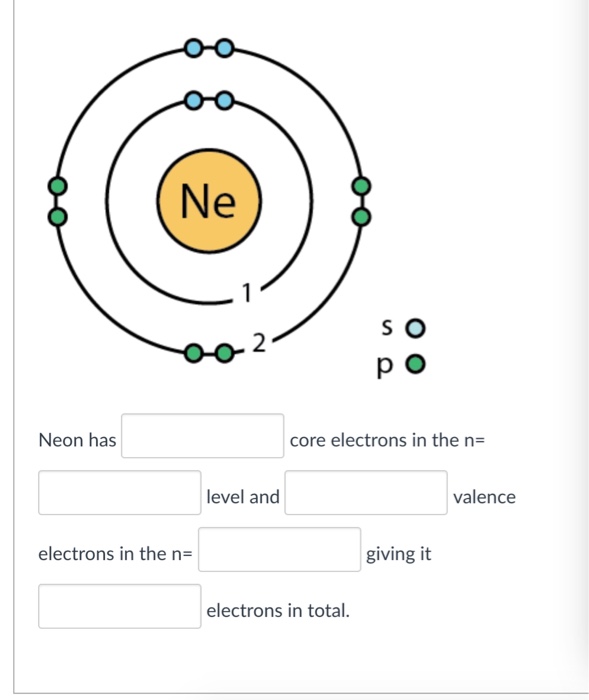
Solved Ne 2 p o Neon has core electrons in the n level and
Therefore, neon has 8 valence electrons. Answer b Calcium has electrons in the first, second, third, and fourth energy levels, as indicated by the leading red 1, 2 's, 3 's, and 4, respectively. Valence electrons are those found in the highest occupied energy level.
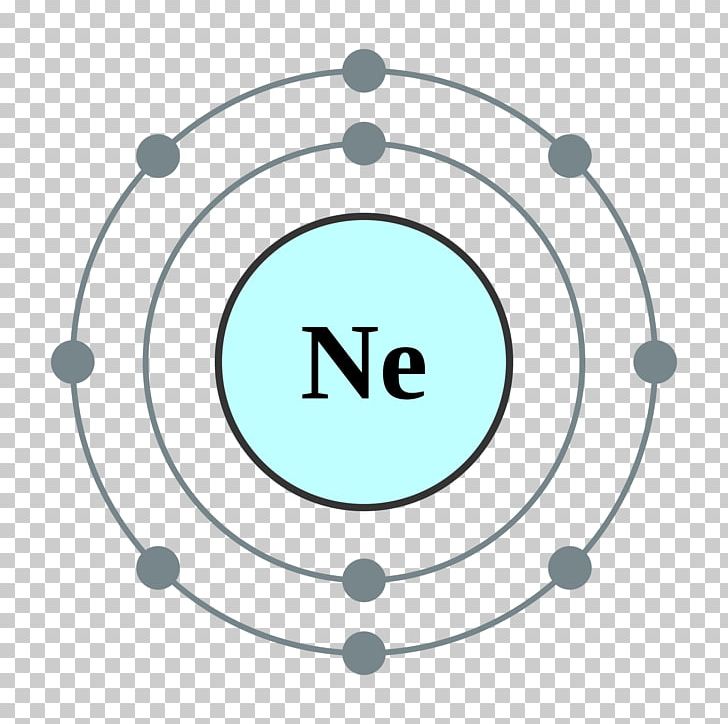
Neon atomic structure Trosadd
1). You can effortlessly find every single detail about the elements from this single Interactive Periodic table. 2). You will get the detailed information about the periodic table which will convert a newbie into pro. 3). You will also get the HD images of the Periodic table (for FREE).

Neon Protons, Neutrons, Electrons Complete Guide
Manahil Ahmed 9 years ago If helium is in group 8 why does it not have 8 valence electrons . And when two hydrogen atoms combine they make a molecule and then have a total of 2 valence electrons if they are then stable then why are the elements in group 2 not stable with 2 valence electrons ? • Comment ( 13 votes) Upvote Downvote Flag Just Keith
silvercraftdesignstudios How Many Valence Electrons Does Antimony Have
Get ready to find out how many valence electrons neon has! In this video, we'll explore the fascinating world of chemistry and discover why valence electrons.