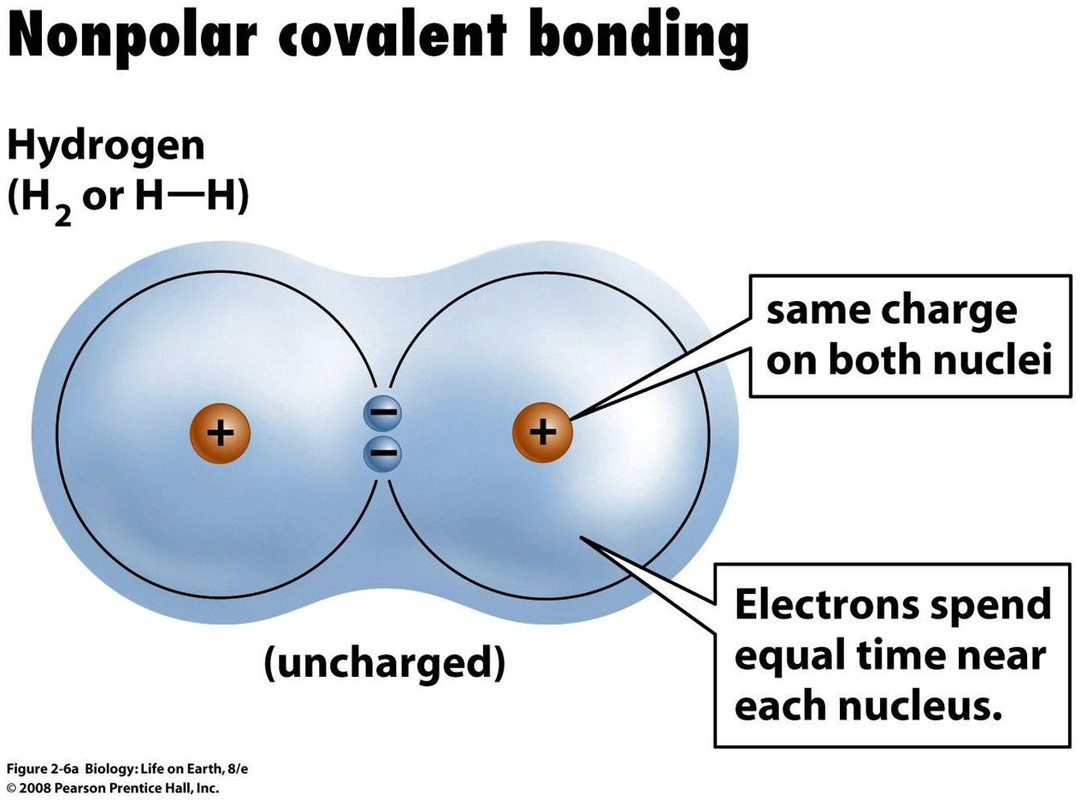
How does a polar covalent bond differ from an nonpolar? Socratic
To understand the polarity of NaCl (NaCl polar or nonpolar), we must delve into the concept of electronegativity - the tugging force with which an atom seeks to pull electrons towards itself in a chemical bond. In the case of NaCl, this electronegativity dance is a spectacle to behold.

is sodium chloride polar or nonpolar
Learn to determine if NaCl (Sodium chloride) is ionic, polar, non-polar based on the Lewis Structure and the molecular geometry (shape).We start with the Lew.
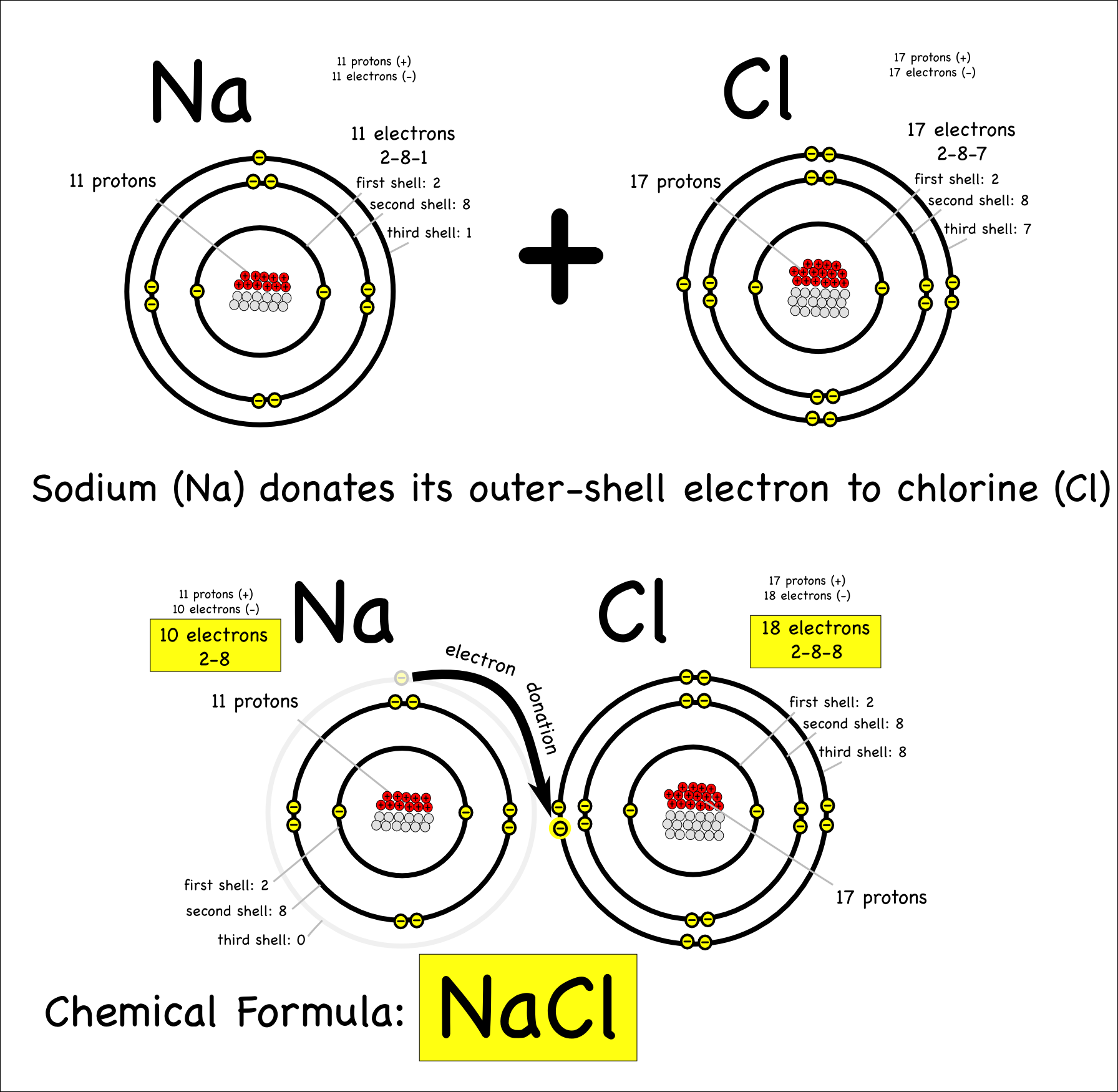
Introducing Covalent Bonding Montessori Muddle
Hi Guys, welcome back to our channel.Today in this video, we help you determine the polarity of the NaCl compound- also known as Sodium Chloride. Unlike othe.

NaCl Molecular Geometry Science Education and Tutorials
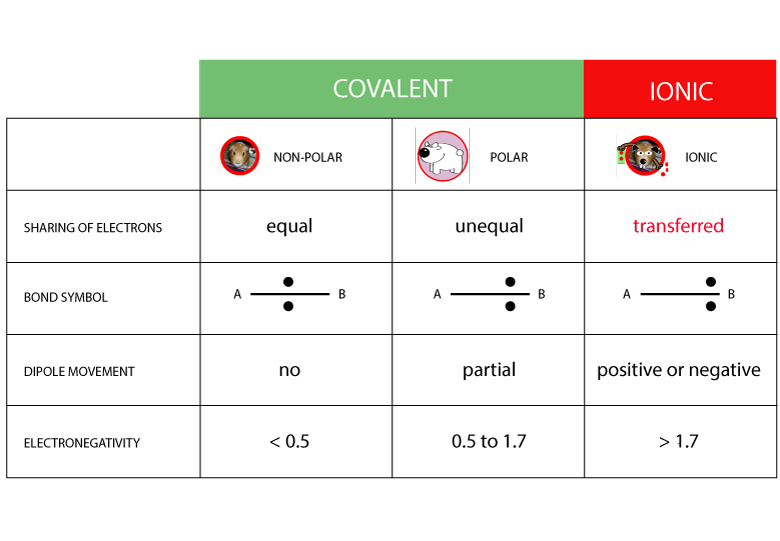
Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. Electronegativity is a measure of the tendency of an atom to attract electrons (or electron density) towards itself.. Likewise, the Na and Cl atoms in NaCl have an electronegativity difference of 2.1, and the Mn and I atoms.
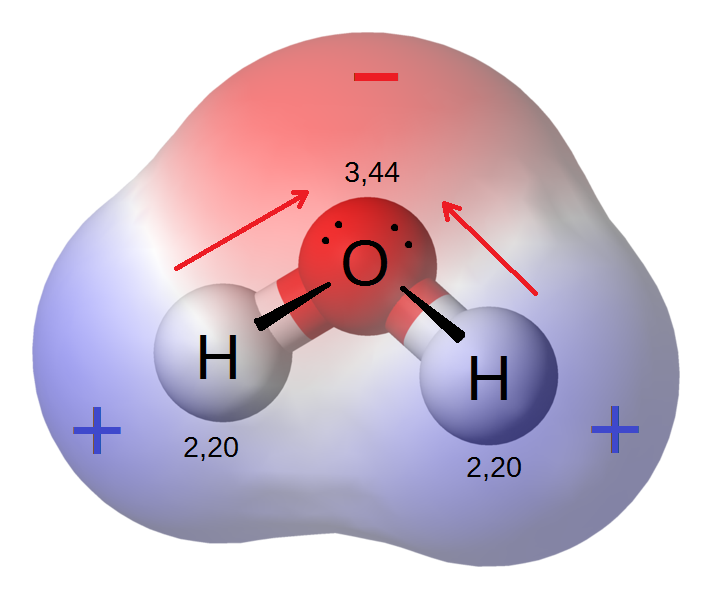
How Do Polar and Nonpolar Molecules Interact With Each Other
Sodium chloride is an ionic compound with a 1:1 ratio of sodium and chloride ions. Its other name is salt. It is found in the oceans and seas and can also be found in the form of rock salt. NaCl makes up between 1% and 5% of seawater. It is a white crystalline solid. It has a molecular weight of 58.44g/mol. Table of Contents
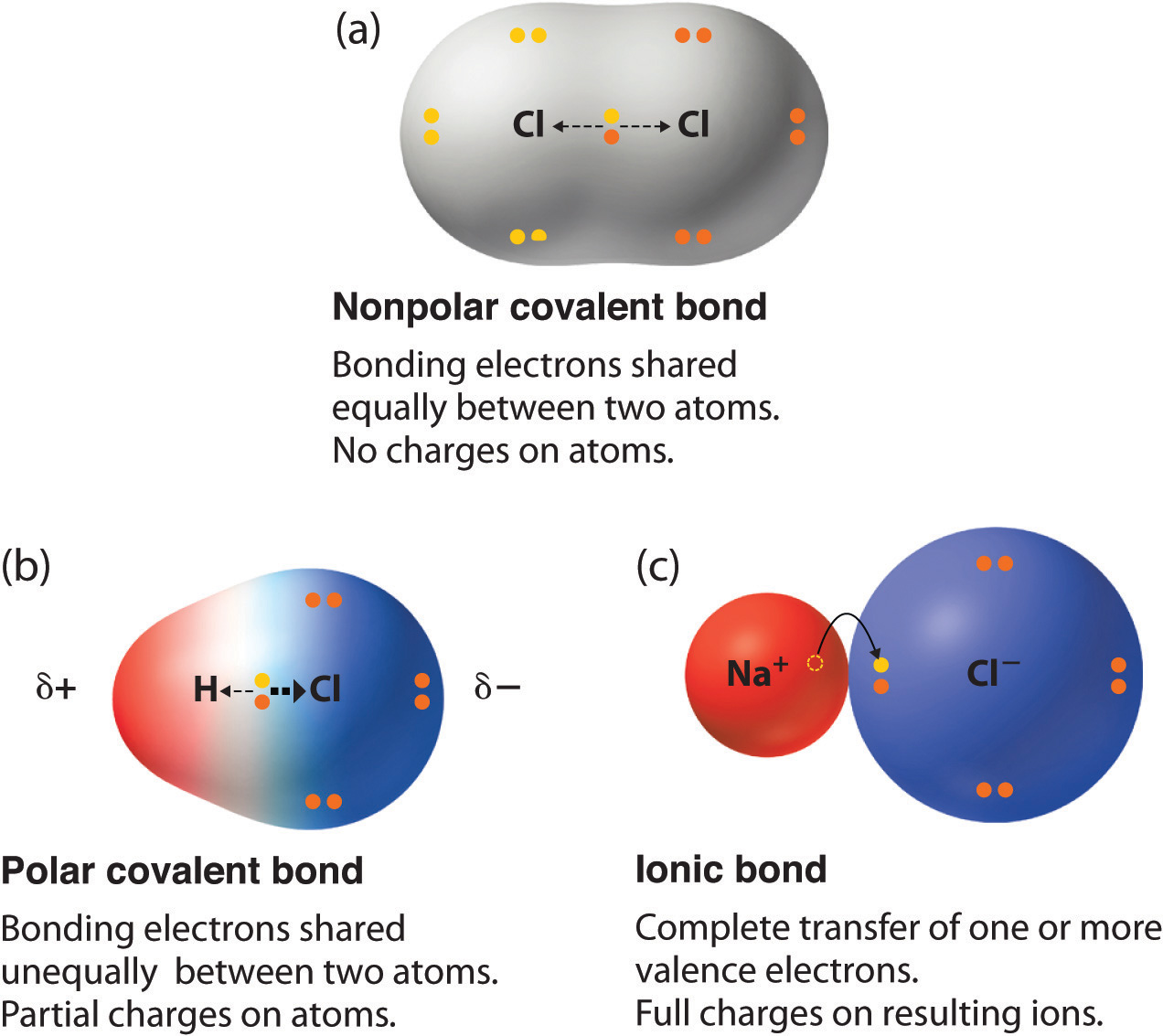
Polar Covalent Bonds
So, NaCl—a very polar substance because it is composed of ions—dissolves in water, which is very polar, but not in oil, which is generally nonpolar. Nonpolar wax dissolves in nonpolar hexane, but not in polar water.. (C 8 H 18), a non-polar organic compound; Solution. Because water is polar, substances that are polar or ionic will.
Comparision of Bonds Surfguppy Chemistry made easy for visual learners
1. How do you determine if the NaCl Lewis structure is polar or nonpolar? The NaCl Lewis structure is nonpolar because it consists of ionic bonds between sodium (Na) and chloride (Cl) ions. Ionic compounds do not have a distinct positive or negative end, resulting in a nonpolar molecule. 2. How can I balance the equation Na + Cl2 → NaCl?
PPT Chemical Bonding I Lewis Theory PowerPoint Presentation, free
Solution Sodium chloride NaCl is polar in nature. Polar molecules are those which can dissolve in a polar solvent. Sodium chloride is an ionic compound. Polar molecules are generally ionic in nature whereas non-polar molecules are dissolved in a non-polar solvent and are generally non-ionic in nature.
PPT Bonding PowerPoint Presentation ID3050946
Whether a bond is nonpolar or polar covalent is determined by a property of the bonding atoms called electronegativity. Electronegativity is a measure of the tendency of an atom to attract electrons (or electron density) towards itself.. Likewise, the Na and Cl atoms in NaCl have an electronegativity difference of 2.1, and the Mn and I atoms.
9.3 Molecular Shape and Molecular Polarity Chemistry LibreTexts
If you stir table salt into water, the crystal lattice of NaCl will begin to dissociate into Na + and Cl − ions. ( Dissociation is just a name for the process in which a compound or molecule breaks apart to form ions.)
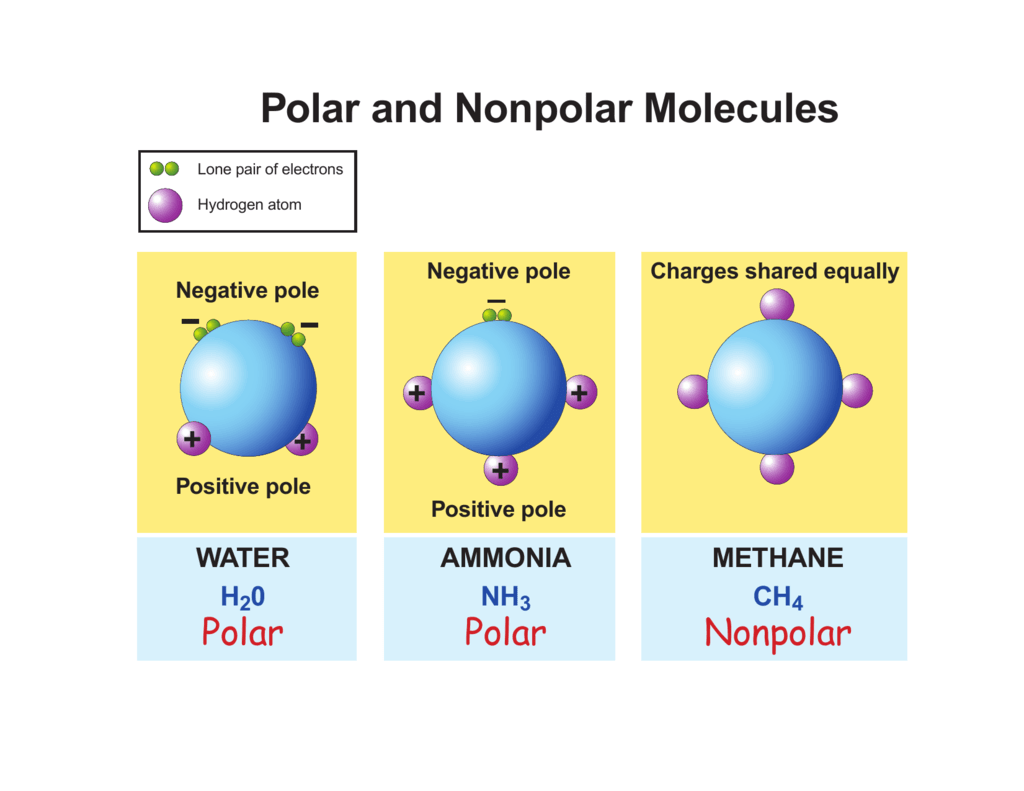
Polar and Nonpolar Molecules
Henry Agnew (UC Davis) 5.10: Electronegativity and Bond Polarity is shared under a not declared license and was authored, remixed, and/or curated by LibreTexts. Covalent bonds can be nonpolar or polar, depending on the electronegativities of the atoms involved. Covalent bonds can be broken if energy is added to a molecule.
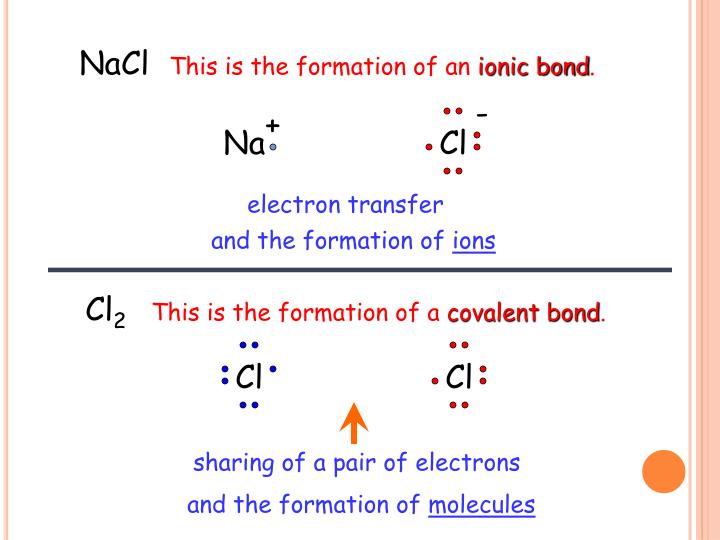
PPT Covalent Bonds Electronegativity differences and ionic/polar
So the difference in electronegativity is somewhere between 1.5 and 2.1, between a polar covalent bond and an ionic bond. So most textbooks we'll see approximately somewhere around 1.7. So if you're higher than 1.7, it's generally considered to be mostly an ionic bond. Lower than 1.7, in the polar covalent range.

NaCl Polar or nonpolar (Sodium Chloride) Chemistry 10, Molecules
Written by Priyanka in Science To answer your question in one word, yes, NaCl or Sodium Chloride is a polar molecule. Wondering how? Read this blog post to know we determined the polarity of the molecule. NaCl is a chemical formula for Sodium Chloride, and it is made of one Sodium atom and one chlorine atom.

Is NaCl Polar or Nonpolar? YouTube
NaCl: Classifying it as polar or non-polar. NaCl is a polar molecule due to the significant electronegativity difference between sodium and chloride ions. The polar nature of the molecule arises from the unequal sharing of electrons between the two ions, resulting in an overall dipole moment. Applications of NaCl's polarity classification
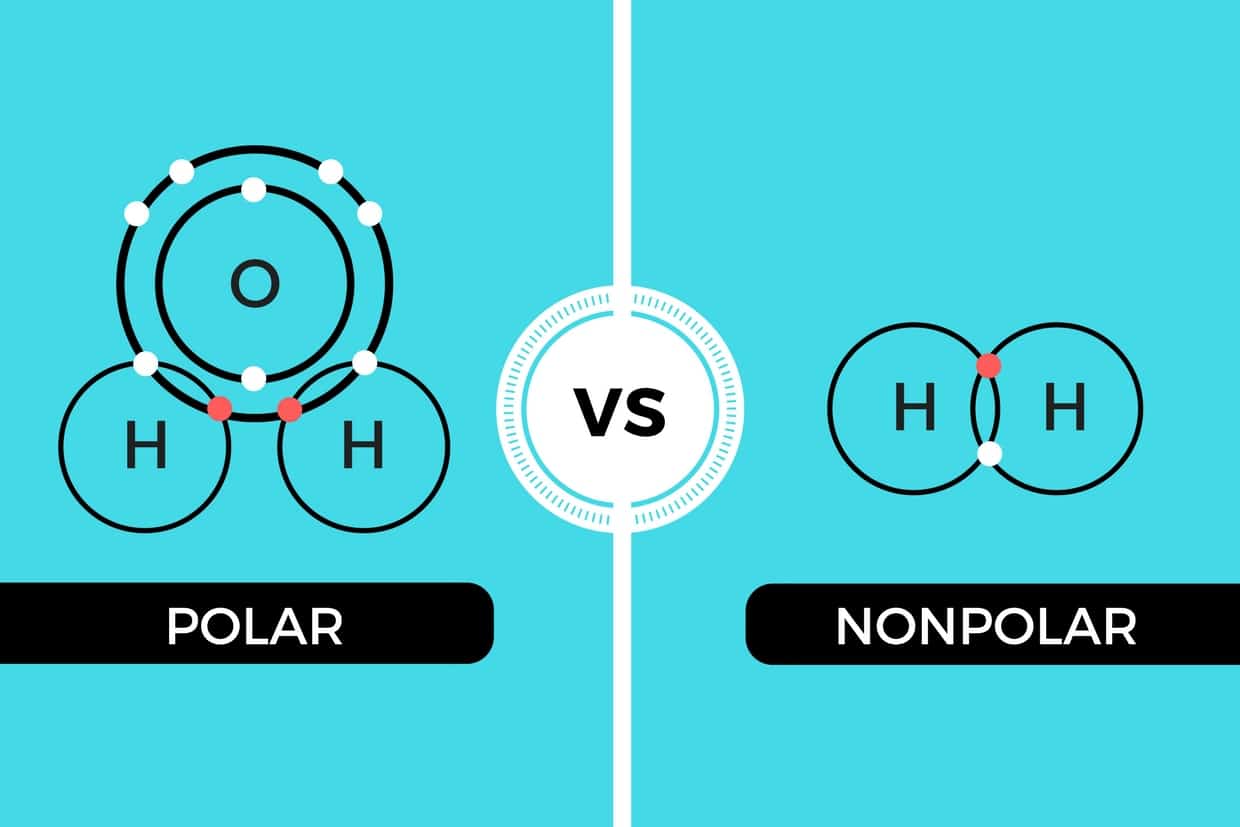
Polar and Nonpolar Covalent Bonds Characteristics & Differences
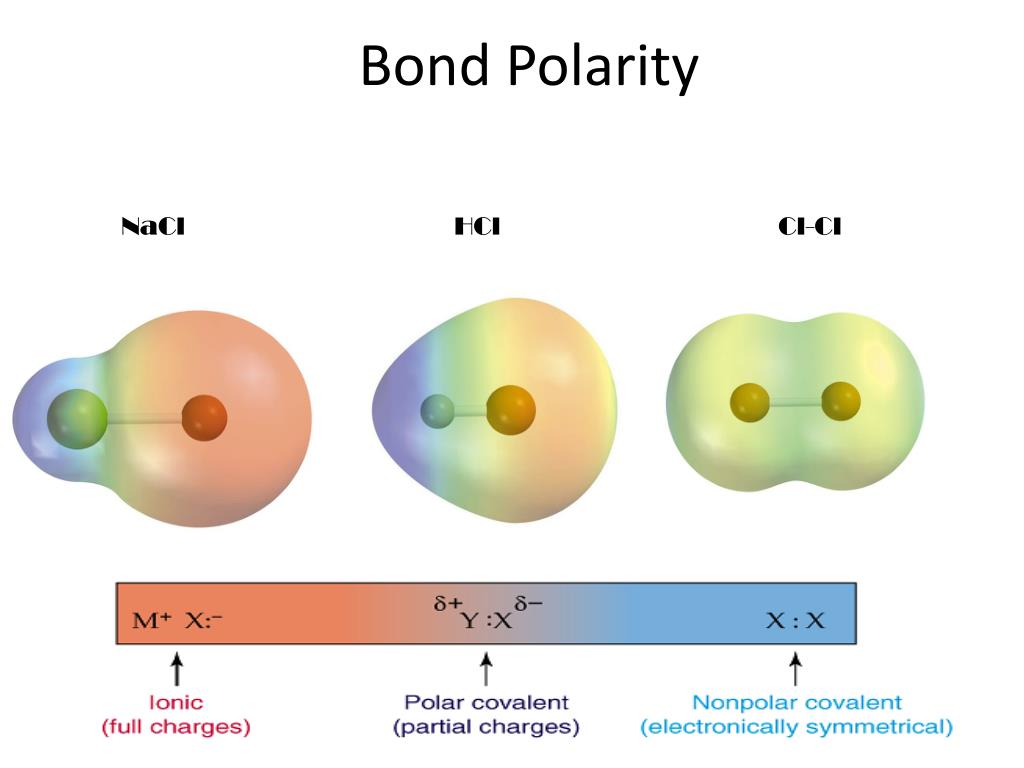
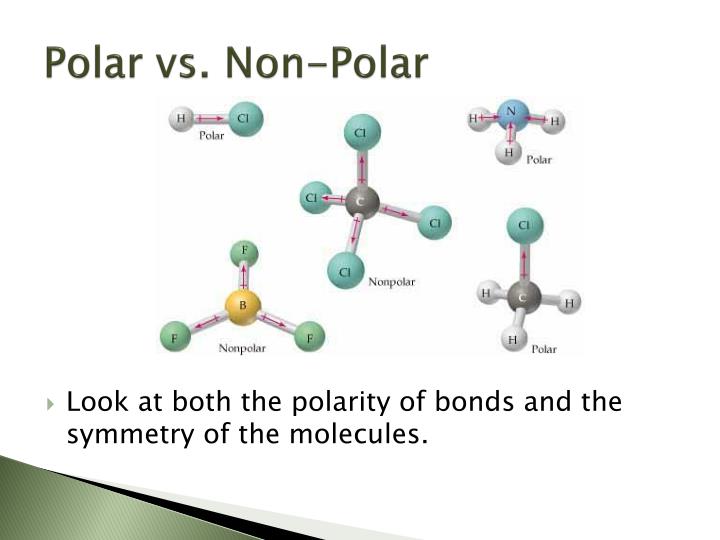
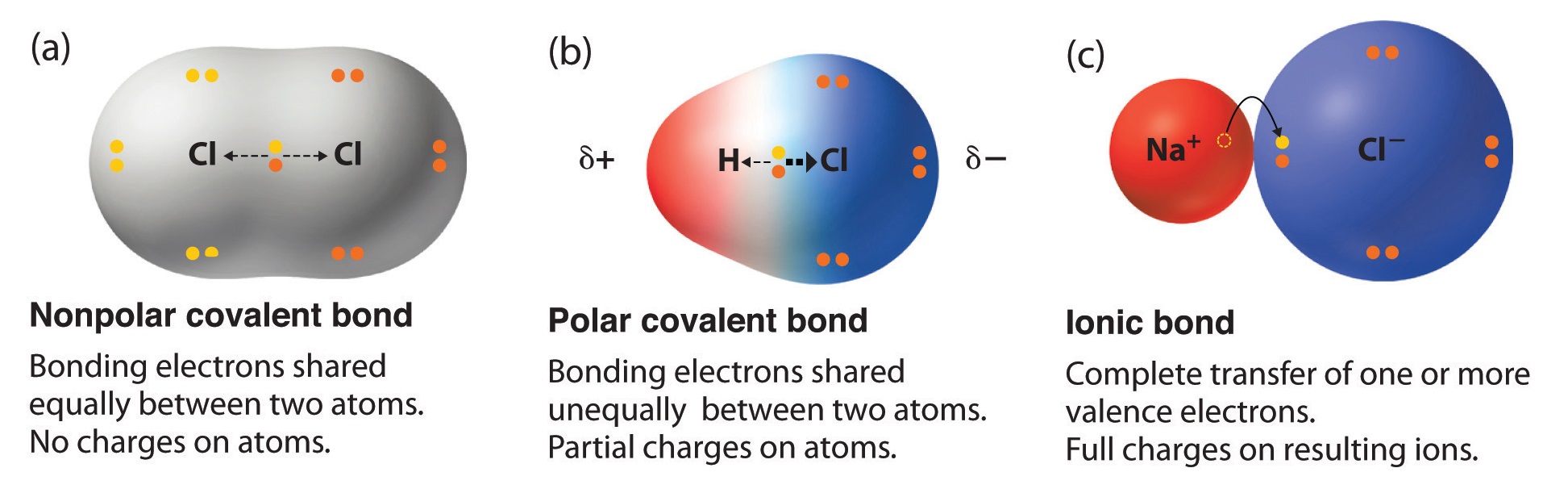
There are two basic types of covalent bonds: polar and nonpolar. In a polar covalent bond, the electrons are unequally shared by the atoms and spend more time close to one atom than the other. Because of the unequal distribution of electrons between the atoms of different elements, slightly positive (δ+) and slightly negative (δ-) charges.
Difference between polar and nonpolar examples
Note ionic compounds, such as sodium chloride (NaCl), are polar. However, most of the time when people talk about "polar molecules" they mean "polar covalent molecules" and not all types of compounds with polarity! When referring to compound polarity, it's best to avoid confusion and call them nonpolar, polar covalent, and ionic.